
[
] 48
chronic hepatitis B infection (positive for HBsAg and
HBeAg) become infected by HBV, and roughly 90 per
cent of these children will turn into chronic carriers. For
those children born from mothers who are chronic carri-
ers (positive HBsAg), approximately 20 per cent will
become infected by HBV and roughly 90 per cent of
them will become chronic HBV carriers. As approxi-
mately 25 per cent of chronic carriers generate serious
diseases such as cirrhosis or hepatic cancer, vaccination
of newborns to protect them from getting chronic HBV
infection is of utmost importance.
The initiation of the Expanded Programme on
Immunization by the World Health Organization
(WHO) and the creation of the Global Alliance for
Vaccines and Immunization (GAVI) – a grand coali-
tion including WHO, UNICEF, PAHO, the Gates
Foundation, Rockefeller Foundation, vaccine indus-
tries non-governmental organizations and many more
– has helped in the battle against hepatitis B.
Currently, in more than 100 countries, especially in
highly endemic areas of Southeast Asia, China, the
Pacific, the Middle East, Sub-Saharan Africa and India,
children are vaccinated against hepatitis B, mostly
using recombinant subunit HBV vaccine. The initia-
tives of the Developing Country Vaccine Industry in
alliance with GAVI and its partners to provide a consis-
tent and sustainable supply of affordable vaccines,
recognizing the essential role of developing country
vaccine producers, may contribute greatly to the
control of the worldwide medical and economic
burdens of HBV infection.
antigenic characteristics that may lead to Major Histocompatibility
Complex (MHC) linked non-responsiveness in some individuals.
Development of novel HBV vaccines to combat
non-responsiveness and increase convenience
In an attempt to overcome non-responsiveness towards the recom-
binant HBV subunit vaccine, a variety of adjuvants including
unmethylated CpG motif, adenovirus and vaccinia virus vectors,
and virosomal technology are being explored. One of the most
promising approaches is the use of vaccines including an S protein-
CpG motif conjugate capable of inducing a T helper
cell-independent immune response against HBsAg, thereby bypass-
ing class II MHC restriction. Indeed, during clinical trials this
innovative recombinant subunit HBV vaccine showed a 100 per cent
seroprotection rate even in seronegative seniors aged 40 to 70 years.
These findings indicate that the CpG-conjugated HBV vaccine is
effective in non-responders to alum-adjuvanted HepB vaccine and
induces a better immune response in immunocompromised indi-
viduals. Moreover, development of efficient drug delivery systems
such as sugarglass, skin patches that employ lymphotoxin beta, and
modified dextran microparticles, may contribute to increased effi-
cacy and improved quality of the HBsAg vaccine. Hence, it is
believed that the introduction of the aforementioned newly-devel-
oped technologies to existing HBsAg vaccines not only increases
efficacy but contributes to convenience and a decrease in the cost
of goods, while enabling the production of HBsAg vaccines with
improved safety and stability.
Supranational measures required for HBV prophylaxis in
developing countries
Between 70 and 90 per cent of infants born from mothers with active
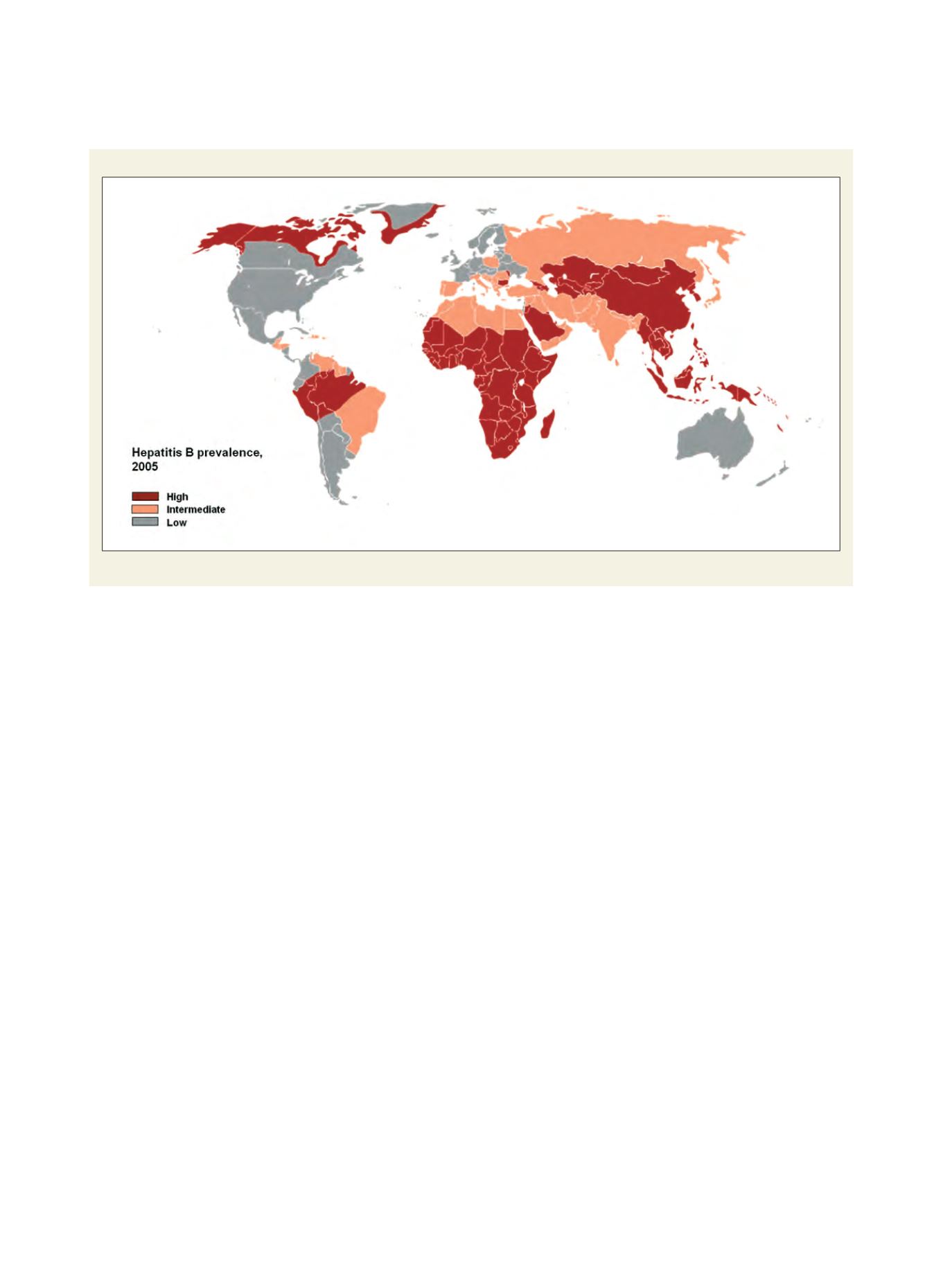
Hepatitis B prevalence
Prevalence of chronic infection with hepatitis B virus
Source: Adapted from: CDC
http://wwwn.cdc.gov/travel/yellowBookCh4-HepB.aspx















