
[
] 80
To develop a successful pandemic flu vaccine R&D
programme and vaccine industry in Taiwan, the task-
forces understand that we need all parties (academic,
government and private industries) to work together
with risk/profit sharing in mind. The NHRI Vaccine
Center plays the transitional roles including pilot plants,
cGMP-grade clinical materials production and cGMP
quality management training, and initiates phase 1 and
2 clinical trials with Investigational New Drug (IND)
submissions. Private industries invest to build manu-
facturing plants and take proven vaccine candidates to
market by conducting phase 3 trials and developing
global marketing strategies. Last but not least, the task-
forces understand that government must provide
long-term vaccine purchase policy and incentive pack-
ages. The three-year short-term R&D flu vaccine
programme was initiated in 2006.
There follow some progress reports on the roles and
achievements accomplished by this R&D programme to
date.
Epidemiology
In order to ensure the creditability and critical mass to
operate efficient surveillance systems for identifying
novel influenza viruses and unusual clusters of respira-
tory infections in humans, an Influenza Epidemiology
Steering Committee, headed by Dr Mei-Shang Ho of
will certainly play important roles in fighting the death rate, but
pandemic flu vaccines must be available as soon as possible. Since
we still do not understand the mechanisms by which pandemic
influenza strains originate, the manufacturers currently do not know
what the compositions of the vaccines should be, and cannot commit
themselves to massive stockpiling of future vaccines. There is now
capacity to manufacture about one billion doses of avian flu vaccine,
but globally we need 6-12 billion doses. To minimize the risk of a
shortage of vaccine supplies, the World Health Organization (WHO)
urged that all governments should organize to implement regional flu
vaccine manufacturing plans:
• To initiate and collaborate on a global pandemic flu vaccine
R&D programme
• To assign a current Good Manufacturing Practice (cGMP)
biological plant to develop flu vaccine emergency production
• If possible, to establish regional vaccine self-manufacturing
facilities capable of producing 20 million doses.
To minimize the risk of flu vaccine supplies, the Taiwan Government
has initiated plans:
• To purchase seasonal flu vaccine from different vaccine suppliers
• To establish a detailed competition purchase mechanism to
control the cost of vaccines
• To implement an influenza vaccine self-manufacturing plan
• To initiate a pandemic flu vaccine R&D programme
• To assign NHRI/CDC to develop a flu vaccine production
emergency plan.
Strategies and direction for pandemic flu vaccine R&D in Taiwan
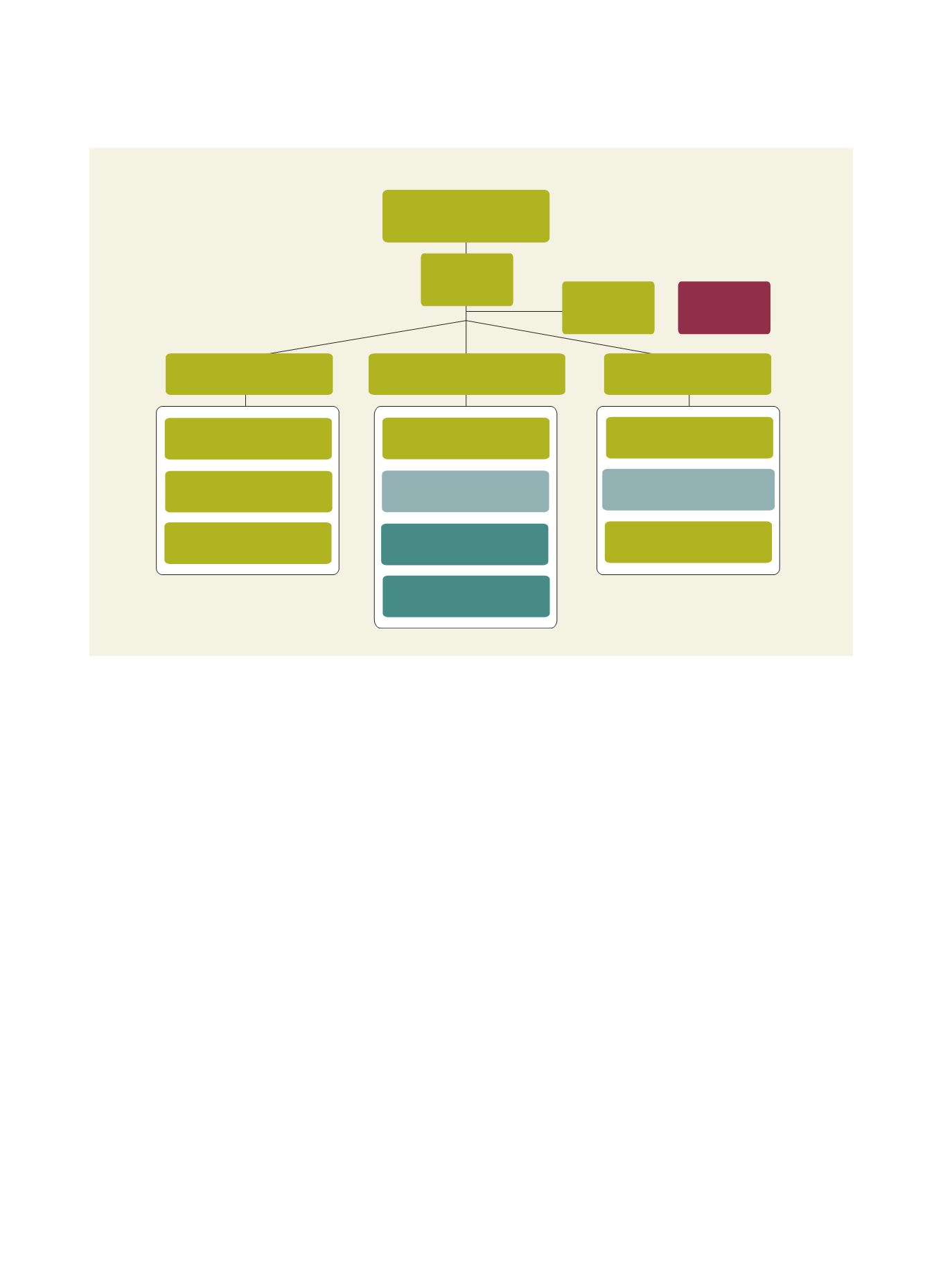
Pandemic flu vaccine
R&D program CDC
Project
management
Steering
committee
Taiwan flu
study groups
Epidemiology arm
Industrializations
Flu vaccine R&D,
manufacturing technologies
Set up efficient diagnostics
surveillance systems
Pandemic flu model studies
Study flu virulence and
antigenic changes
Conduct prototype pandemic
vaccine trials
cGMP quality operation and
management training programs
Strength product license and
registration program
International
collaboration
Novel pandemic flu
vaccine development
Pandemic flu vaccine
emergency production
Establish flu vaccine product
characterization core facility
Source: Taiwan CDC and NHRI
















