
[
] 70
influenza. The lessons learned from research and devel-
opment in the Aids vaccine field will go a long way to help
build this capacity. An example of this is the Biovac
Institute, which has partnered with SAAVI and is also in
talks with the Istituto Superiori di Sanità in Italy to support
its vaccine manufacturing capability. The latter is part of
the Italy-South Africa Programme to support the Ministry
of Health of South Africa in the implementation of a
national programme of global response to HIV and Aids.
Despair among HIV vaccine researchers
While an HIV vaccine remains the primary goal for a
comprehensive strategy to curb the global HIV
pandemic, the path to success is unknown and has
become more complicated. In 1984 Margaret Heckler,
Health and Human Services Secretary under US
President Ronald Reagan, told Americans: “We hope to
have a vaccine ready for testing in about two years.”
This was after scientists had identified HIV as the virus
responsible for Aids.
Originally, vaccine research focused on identification
of immunogens that would elicit neutralizing antibodies
to prevent infection. Two phase-3 trials did not find any
protection against HIV infection. Nonetheless, efforts to
design vaccines that elicit antibody responses continue.
The importance of T-cell immunity in containing HIV
infection was influenced by studies of early infection in
people and experiments on non-human primates.
The results of the recent failed vaccine clinical trial
(Step Study), however, have profoundly affected the HIV
vaccine development field. Participants receiving the
vaccine, who had higher pre-existing levels of adenovirus
absence of a known correlate of protection and the widespread
genetic diversity of the virus pose substantial scientific hurdles, and
this remains one of the greatest challenges in developing an effec-
tive HIV vaccine.
The vision of the African Aids Vaccine Programme (AAVP), an initia-
tive sponsored by the World Health Organization (WHO) and the Joint
United Nations Programme on HIV/Aids (UNAids), is an Aids-free
Africa through an effective vaccine. The AAVP is a network of African
HIV vaccine stakeholders committed to promoting HIV vaccine devel-
opment for Africa through research, advocacy, partnership and
contribution to capacity strengthening and policy development.
In southern Africa, subtype C accounts for over 95 per cent of infec-
tions (19-22). Subtype C is also largely responsible for the epidemics
in Ethiopia and India, and accounts for over 50 per cent of HIV-1
infections globally. In response to the devastating subtype C epidemic
in southern Africa, the South African Aids Vaccine Initiative (SAAVI),
a lead programme of the South African Medical Research Council
(MRC), in collaboration with the University of Cape Town and the US
NIH, developed two subtype C HIV vaccines – SAAVI DNA-C2 and
SAAVI MVA-C – to be employed together in a prime-boost protocol.
In light of the AAVP goals, it is heartening to note that the first
Aids vaccine constructed and developed in Africa is currently being
tested in the US – so much for being dubbed ‘third-world’! This trial
will examine the safety and immunogenicity of the candidate
vaccines, in both a subtype C region (South Africa) and a subtype B
region (US), as part of the scientific agenda of the HIV Vaccine Trials
Network to find vaccines that will prevent HIV infections in adult
and adolescent populations globally.
It is extremely important for developing nations, and especially
Africa, to build the capability of manufacturing vaccines, since it is
unlikely that stockpiles of critical vaccines will be made available to
them should there be a global pandemic of, for example, avian
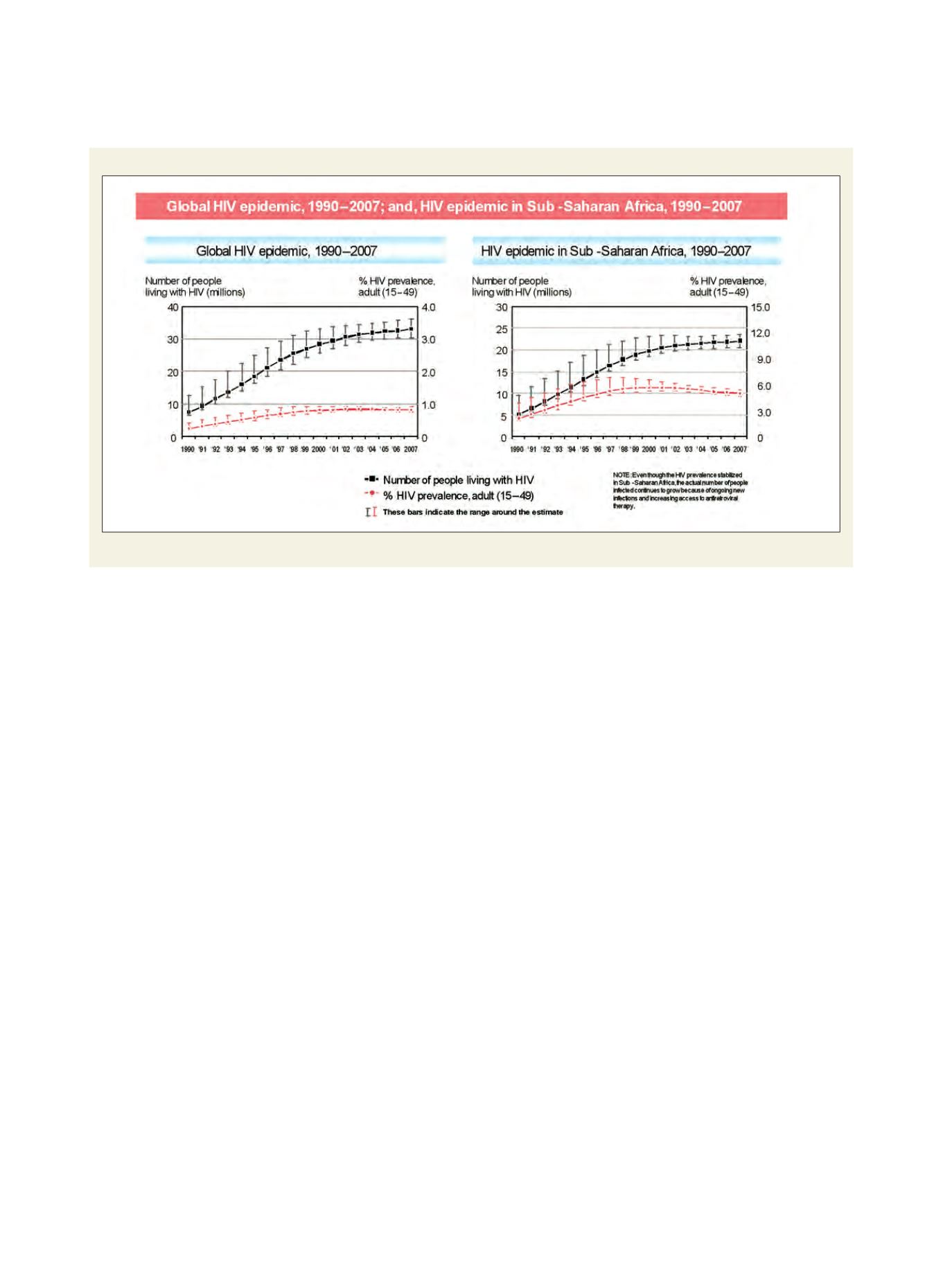
Estimated number of people living with HIV and adult HIV prevalence
Sub-Saharan Africa bears a disproportionate share of the global HIV burden
Source: UNAids Report 2008
















