
[
] 25
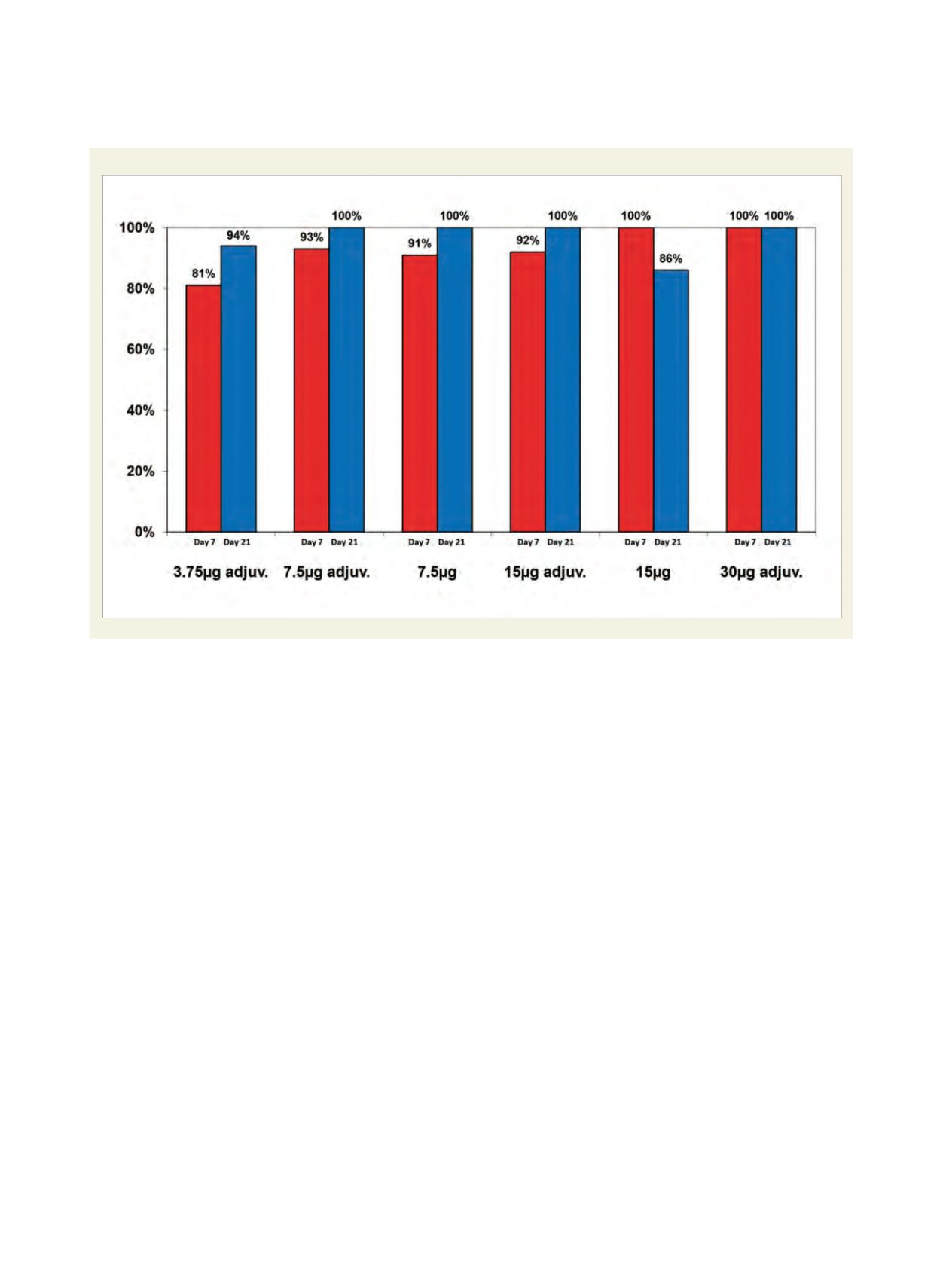
Percentage of subjects with neutralizing antibody response to the booster vaccine (MN titer>20)
Source: Baxter Bioscience
ularly fever, when used as trivalent seasonal vaccines. All clinical
studies to date (on around 4,000 subjects) have demonstrated that
the Vero cell derived whole virus vaccine was well tolerated in adult
and elderly populations. Data from paediatric studies is not yet
available.
The data available from clinical studies confirmed the excellent
immunogenicity seen in pre-clinical animal studies.
6
Clinical studies
carried out with A/Vietnam/1203/2004 or A/Indonesia/05/2005
demonstrated that doses as low as 3.75 micrograms or 7.5 micro-
grams resulted in induction of seroprotective levels of neutralizing
antibody in more than 70 per cent of subjects after two immuniza-
tions. This immunization regimen also resulted in induction of a
substantial cross-reactive antibody response. In these studies, neutral-
izing antibodies were also induced against a clade 0 strain and, to a
lesser extent, to a clade 2.1 strain, after immunization with a clade
1 strain vaccine. With this immunization schedule, the highest
neutralizing antibody response was obtained 42 days after initiation
of immunization and required a two-dose schedule.
However modelling of pandemic spread has shown that induction
of an immune response to a pandemic strain in a significant propor-
tion of the population within two weeks after initiation of an
outbreak is critical in order to interrupt virus transmission.
Therefore, conventional strategies requiring two vaccinations 21 days
apart at the onset of a pandemic are unlikely to result in a rapid
protection of the population. Alternative strategies would be required
to ensure rapid induction of immunity in the population. Such a
strategy would be the concept of pre-pandemic priming immuniza-
tion with an existing available H5N1 strain, which may not
necessarily be a complete match to an emerging H5N1
strain. Boosting would then be carried out with a single
dose of the optimally or closely matched pandemic
strain vaccine.
This concept has been investigated in a study where
subjects were primed with two doses of a Vietnam
(clade 1) strain vaccine in dosages ranging from 3.75
micrograms to 30 micrograms. Formulations with and
without alum adjuvant were included in the priming
immunization regimen. Approximately 18 months after
priming, the subjects were then boosted with one dose
of a 7.5 microgram non-adjuvanted formulation of an
Indonesia strain (clade 2.1) vaccine, as a model for the
pandemic strain vaccine. The collected data demon-
strated that 81 per cent to 100 per cent of the subjects
developed a seroprotective neutralizing antibody titre
to the booster strain vaccine as early as seven days after
the booster immunization. This prime-boost strategy,
which requires only a single dose of vaccine in the
event of a pandemic, would also result in a substantial
increase in the availability of vaccine, in addition to
the more rapid induction of protective immunity in the
population early in a pandemic. The use of Vero cell
technology, which allows more rapid and robust
vaccine supply together with use of such prime-boost
immunization strategies, would have significant
impacts for global public health in the event of an
influenza pandemic.
















